cGMP Manufacturing
and Quality Control
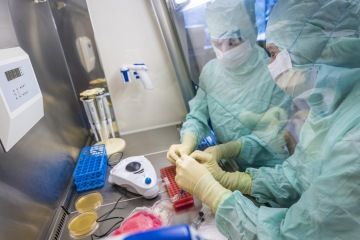
Professional cGMP manufacturing services covering all well known production approaches and QC methods for cell and gene therapy products and viral vectors.
-
We can offer all services covering autologous or allogenic cell & gene therapy products
as well as viral vectors, all within dedicated or shared manufacturing suites. - Manufacturing process from various starting materials (e.g. apheresis products, whole blood, tumor tissue etc.)
- Manufacturing of each batch is managed via internal laboratory software system LaSSo™
- Automated or manual fill and finish services
- Customized cryopreservation of DS and final DP as well as samples for further QC
- Preparation and characterization of cell banks from MCB to WCB
- Labeling and re-labeling services
- QC testing based on variety of cell based and molecular methods
- Microbiology testing
- Rapid sterility testing methods
- Characterization assays and release testing
- Backup for materials and outsourced tests
- All necessary backup systems in place – all for GMP facility, including independent storage
- Stability studies
- Testing of raw materials
- Ability to accommodate Person-in-Plant